Elekta Announces First Patients Treated with Major Breakthrough in Cancer Therapy
St. James’s University Hospital (Leeds, UK) debuts clinical use of Agility multileaf collimator (MLC)
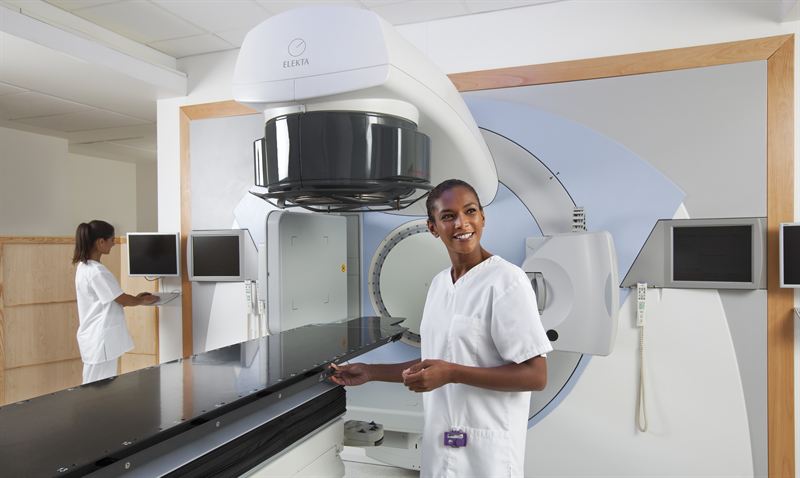
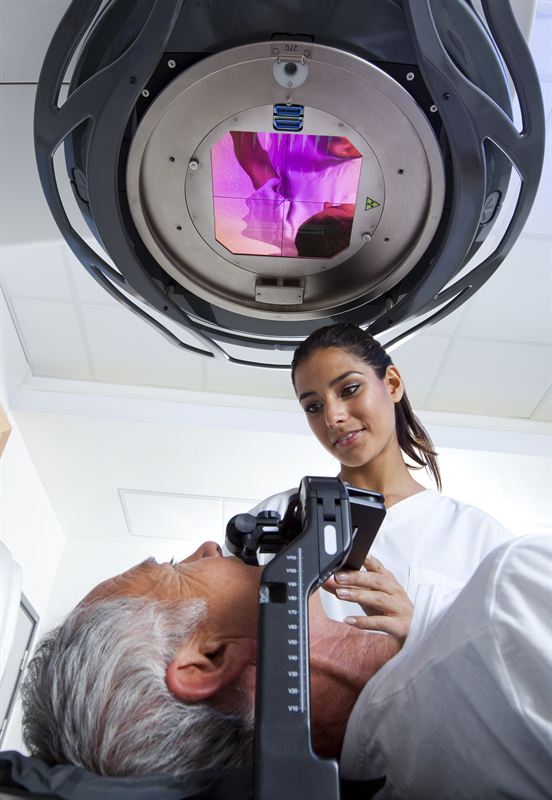
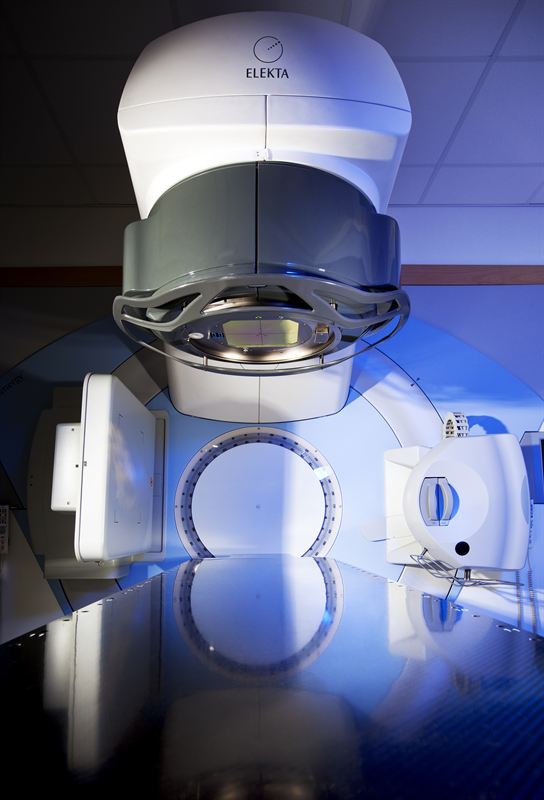
LEEDS, England, April 19 – Using Agility™*, Elekta’s latest MLC innovation, patients at St. James’s University Hospital were the first in the world to benefit from this modern advancement in the treatment of cancer. With twice the number of leaves typical of many standard MLCs, Agility precisely sculpts delivered radiation to the unique contours of the tumor while reducing the risk of exposure to surrounding healthy tissue.
A multileaf collimator, a device made up of numerous individual tungsten leaves, is commonly used to shape beams of radiation as therapeutic doses are delivered from different angles around the patient.
“This truly represents a radical improvement in the way we deliver radiotherapy, combining both speed and precision in tailoring the radiation beams to the exact shape of the patient’s tumor,” says Vivian Cosgrove, Ph.D., head of radiotherapy physics at St. James’s. “Agility enables a faster delivery of advanced radiotherapy treatments. Reduced treatment times will lead to a better experience for the patient and will improve access to the technology for the benefit of more of our patients.”
The product of an extensive R&D effort by Elekta to transform cancer care, the leaves of Agility are also capable of traveling at twice the speed of other MLCs commonly used in radiotherapy. This unique capability supports an added capacity for precision beam shaping as well as shorter treatment times, increasing both patient comfort and the clinic’s delivery efficiency. Moreover, relying on a new and innovative design, Agility MLC has demonstrated extraordinarily low leaf transmission, to reduce the patient’s non-therapeutic radiation exposure.
Recently, Agility was awarded the CE Mark, enabling medical centers across Europe and other regions to adopt the technology for their cancer patients.
“Agility’s versatility combined with the potential patient benefits have generated much enthusiasm and market demand,” says Olof Sandén, Elekta’s Executive Vice President, Europe and AFLAME. “We look forward to many more cancer patients benefiting from this breakthrough technology.”
Learn more at www.elekta.com/agility.
*Agility is not available for sale or distribution in all markets. Please contact your local Elekta representative for details.
For further information, please contact:
Stina Thorman, Vice President Corporate Communications, Elekta AB
Tel: +46 8 587 254 37, email: [email protected]
Time zone: CET: Central European Time
Michelle Joiner, Director, Global Public Relations and Brand Management, Elekta
Tel: +1 770-670-2447, email: [email protected]
Time zone: ET: Eastern Time
Johan Andersson Melbi, Investor Relations Manager, Elekta AB
Tel: +46 702 100 451, email: [email protected]
Time zone: CET: Central European Time
The above information is such that Elekta AB (publ) shall make public in accordance with the Securities Market Act and/or the Financial Instruments Trading Act. The information was published at 07:30 on April 19, 2012.
About Elekta
Elekta is a human care company pioneering significant innovations and clinical solutions for treating cancer and brain disorders. The company develops sophisticated, state-of-the-art tools and treatment planning systems for radiation therapy, radiosurgery and brachytherapy, as well as workflow enhancing software systems across the spectrum of cancer care. Stretching the boundaries of science and technology, providing intelligent and resource-efficient solutions that offer confidence to both healthcare providers and patients, Elekta aims to improve, prolong and even save patient lives.
Today, Elekta solutions in oncology and neurosurgery are used in over 6,000 hospitals worldwide. Elekta employs around 3,300 employees globally. The corporate headquarters is located in Stockholm, Sweden, and the company is listed on the Nordic Exchange under the ticker EKTAb. Website: www.elekta.com.